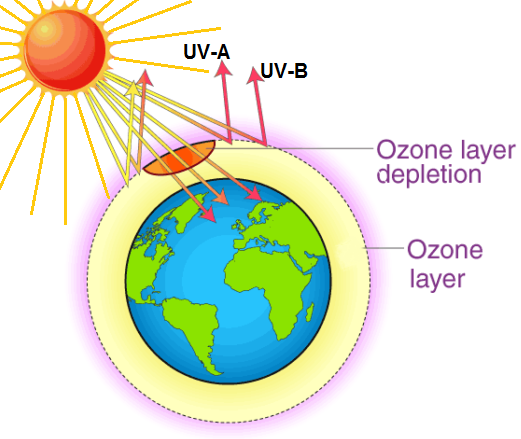
Ozone is a highly reactive trioxygen gas (O3). About 90% of ozone is found in the stratosphere, about 10 to 40 km above the Earth’s surface. The stratospheric region where the highest ozone concentration occurs is known as the ozone layer. It extends over the entire globe with some variation in altitude and thickness. The remaining ozone is found between the ground and the stratosphere.
Depending on where it is in the atmosphere, ozone affects life on Earth in either good or bad ways. The ozone layer filters out harmful ultraviolet radiation (ultraviolet-B, UVB), which is associated with the occurrence of skin cancer (malignant melanoma) and cataracts, reduced agricultural productivity, and disruption of marine ecosystems.
On the positive side, ultraviolet exposure can suppress the clinical symptoms of multiple sclerosis independently of vitamin-D synthesis. Furthermore, UV generates nitric oxide, which may reduce blood pressure and generally improve cardiovascular health. UV exposure leads to the release of endorphins that are linked to both pain relief and euphoric feelings.
Since the 1970s, scientists have pushed for the regulation of chlorofluorocarbons (CFCs), chemicals found in daily-use items like air conditioners, refrigerators, and aerosol sprays, due to their destructive effects on ozone. The evidence for the ozone depletion came from the scientists' observations that showed a dramatic drop in the ozone concentration over Antarctica since the 1970s. This discovery, which was published in Nature in 1985, set the world into action to regulate the use of ozone-depleting chemicals.
The leading causes of ozone variability in the upper stratosphere are photochemical production and destruction. But when it comes to the lower stratosphere, dynamic processes like wind and temperature changes are the leading causes of changes in ozone concentration since the lifetime of ozone in the stratosphere is larger than the timescale of transport. Human activities like the combustion of fossil fuels, biomass burning releases nitrogen oxides and volatile organic carbon that generates ozone in the troposphere which is called “evil” ozone.
The Montreal Protocol is the first multilateral environmental protocol signed in 1987 by all UN member nations to phase out harmful substances, such as CFCs, hydrochlorofluorocarbons, freons, and halons, commonly used in refrigerants, propellants, and solvents. The treaty has primarily been considered a great accomplishment, with global mean total ozone recovery in 2020 to pre-1980 levels.
As a reminder for preserving the ozone layer, the UN General Assembly, on January 23, 1995, adopted resolution 49/114, which proclaims September 16 as World Ozone Day. The celebration is a reminder about the signing of the Montreal Protocol – a framework to control total global production and consumption of substances that deplete ozone. The ultimate objective is to eliminate those substances based on developments in scientific knowledge and technological information.
The Ozone Hole over Antarctica
Winds continuously stir the Earth's atmosphere. So, the ozone-depleting gases get mixed throughout the atmosphere. These gases, which are insoluble in water and relatively unreactive, get mixed throughout the lower atmosphere. When the air rises from the lower atmosphere into the stratosphere, mainly in tropical latitudes, the winds move this air poleward both north and south from the tropics so that air throughout the global stratosphere contains nearly equal amounts of chlorine and bromine.
In the Southern Hemisphere, the massive landmass of Antarctica is surrounded by the ocean. This symmetry allows a frigid region in the stratosphere in winter over Antarctica, isolated by a band of strong winds circulating around that region. Very low stratospheric temperatures promote the formation of clouds responsible for chemical changes that stimulate chemically active chlorine and bromine production, leading to rapid ozone loss when sunlight returns to Antarctica in September and October every year. About 1,00,000 ozone molecules will be destroyed by one chlorine atom before it is eliminated from the stratosphere. This results in the Antarctic ozone hole. Similar conditions do not exist over the Arctic. The wintertime temperatures in the Arctic stratosphere are not persistently low for as many weeks as over Antarctica, which results in correspondingly less ozone depletion there.
Three decades later
According to the World Meteorological Organization press release in January 2021, the Antarctic ozone hole – one of the deepest and the largest ozone layer gaps in the last 36 years – has closed, thanks to the Montreal Protocol. The success of the Montreal Protocol prompted a wave of crucial follow-up agreements, including the Copenhagen Amendments in 1992. The original protocol, combined with the revision, has levelled off the destruction of the ozone. Even though the progress made in reducing the area of the hole has been nominal, there is a massive environmental success. Allowing the ozone hole to increase would have cost many lives around the world. By mitigating the damage and allowing the painfully slow recovery to begin, the timetable in the Montreal Protocol has succeeded in its goal of reducing the destructive effects of ozone depletion.
A small effort from each of us can contribute to preserving the ozone. When you buy an AC and fridge, make sure that you go in for the brand that has lower Global Warming Potential (GWP) and uses zero Ozone Depletion Potential or eco-friendly refrigerant in their products. Let us work to heal the ozone hole and forge a brighter and more equitable future for all humanity.
(The author is a Scientist working on climate change topics, and a freelance writer)